Research & Development
Kimera focuses on developing protein-based treatments for metabolic disorders in which the body cannot properly clear certain molecules. Many metabolic diseases occur when natural biological pathways fail to remove or process specific substances. As a result, harmful molecules build up in the blood or tissues over time, disrupting the body’s normal balance. Current treatments often try to reduce the production of these substances or partly compensate for the defect, but they may not fully restore normal function. Our approach is to design engineered fusion proteins that help the body remove these harmful molecules by redirecting them through alternative, naturally existing cellular clearance pathways.
Technology
Kimera’s platform is based on engineered chimeric proteins that combine two key functions in a single molecule. A binding domain that specifically recognizes the disease-causing substrate and a trafficking domain that directs the substrate to an efficient alternative cellular clearance pathway. By linking these two functions, the technology bypasses defective biological systems and promotes receptor-mediated uptake of harmful molecules, helping restore physiological balance. The platform is modular and adaptable. Binding domains can be selected to target different metabolic substrates, while trafficking modules can be tailored to specific receptors and tissues. This flexible design enables rapid development of mechanism-based therapies across multiple metabolic disorders.
Kimeragene Therapy
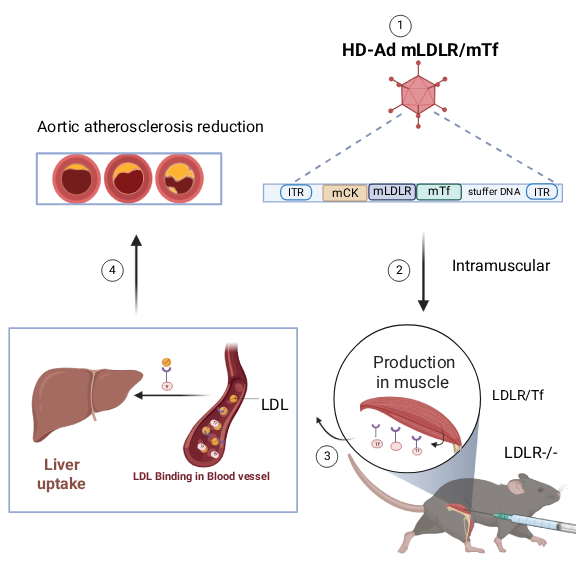
Kimeragene is a gene therapy designed to restore physiological LDL clearance in patients with familial hypercholesterolemia, including those with severely impaired LDL receptor (LDLR) function. The therapy delivers a genetic construct encoding a proprietary chimeric fusion protein. The therapeutic gene is administered using a helper-dependent adenoviral vector engineered for efficient gene transfer and reduced immunogenicity. Expression is driven by a muscle-specific promoter to enable sustained systemic production of the therapeutic protein.
Mechanism of Action
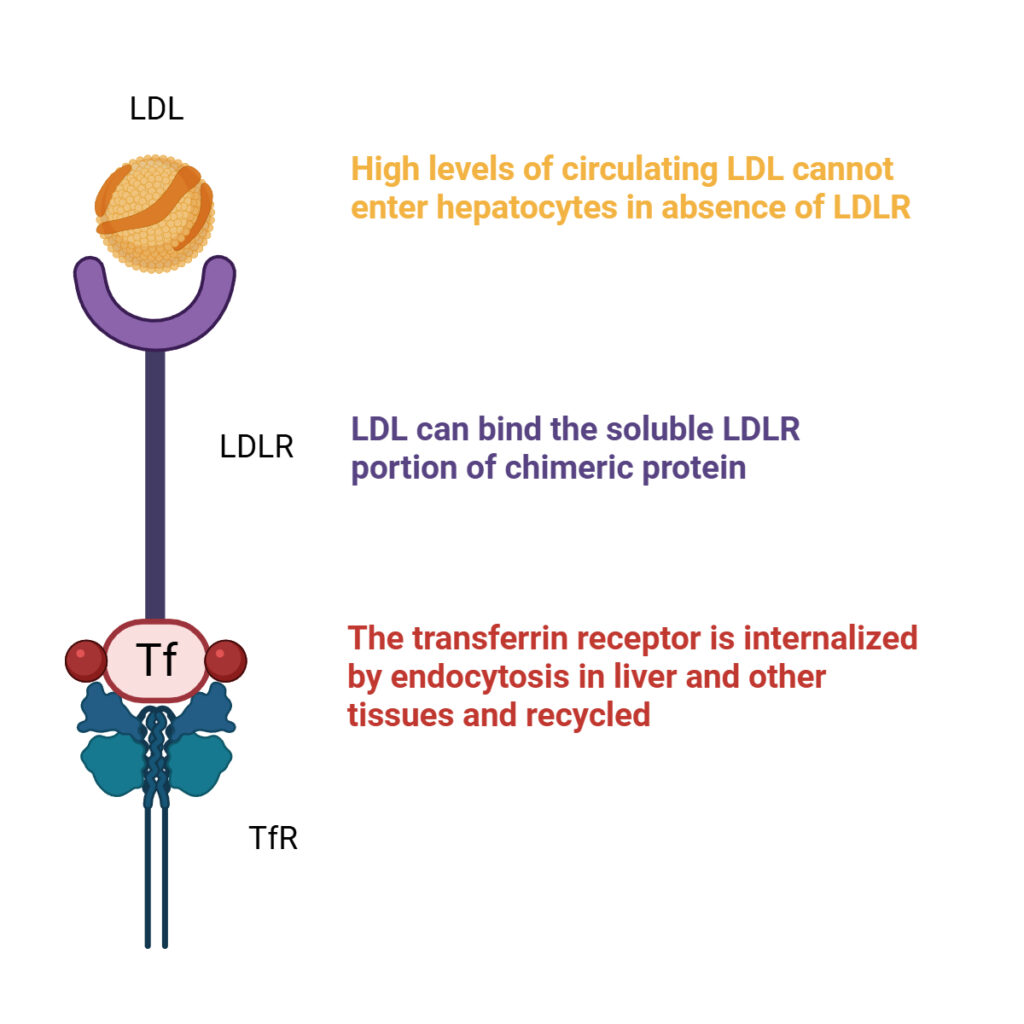
Kimeragene is based on a functional bypass strategy. The engineered fusion protein combines the extracellular domain of LDLR with transferrin. This design enables binding of circulating LDL particles and their internalization through transferrin receptor (TfR)-mediated uptake.
Intramuscular administration of a therapeutic vector in the LDLR-deficient preclinical model leads to local production of the engineered fusion protein in muscle tissue.
By redirecting LDL particles to a naturally active receptor system, Kimeragene restores LDL internalization and clearance even when endogenous LDLR activity is absent or severely reduced.
Preclinical Results
In LDLR-deficient preclinical models, a single administration of Kimeragene resulted in:
- Restoration of LDL uptake
- Significant and durable reduction in circulating LDL-C levels
- Reduction in aortic atherosclerotic burden
- Favorable safety profile with no major systemic adverse effects
These findings provide proof-of-concept for a novel, mechanism-driven therapeutic approach aimed at addressing the high unmet medical need in severe familial hypercholesterolemia.